Chemistry Review Resources
Answers to Practice Test
|
11. A 17. D
12. D 18. A 13. B 19. C 14. D 20. C 15. D 16. B |
| ||||||
Sorting Matter
Chemical vs. Physical ChangeProperties of Metals and Non MetalsAtomic Theory HistoryCreating Ionic CompoundsChemical Word Reactions |
Chemical or Physical ChangeMetals, Nonmetals, CompoundsReaction Rate
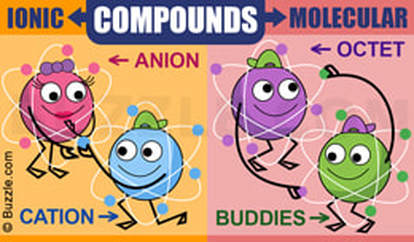
Build an AtomParts of an atomIonic vs MolecularChemical Naming | ||||||
Naming Notes
Molecular or Covalent CompoundsMolecular or Covalent Compounds have specific properties:
- They are between at least 2 non metals. - They share electrons - Usually have low melting and low boiling points - Tend to be soft - Not very good conductors Ionic Compounds
| |||||||
Ionic compounds have specific properties:
- They are between a metal and a non metal.
- They transfer electrons
- The charges of the ions must balance (Na ion has a charge of 1+, Cl ion has a charge of 1-. Therefore the formula is NaCl)
- Have high melting and high boiling points
- Conductive when liquid
- Usually soluble
- They are between a metal and a non metal.
- They transfer electrons
- The charges of the ions must balance (Na ion has a charge of 1+, Cl ion has a charge of 1-. Therefore the formula is NaCl)
- Have high melting and high boiling points
- Conductive when liquid
- Usually soluble
What we know about the Periodic Table
|
Atomic Number: the number of electrons, the number of protons
Atomic Mass: The mass of the atom Neutrons: To find the number of neutrons, you subtract the number of protons from the atomic mass and round to the nearest whole number. Groups= columns in the periodic table Periods= rows in the periodic table Group 1 is known as the Alkali Metals
|
|
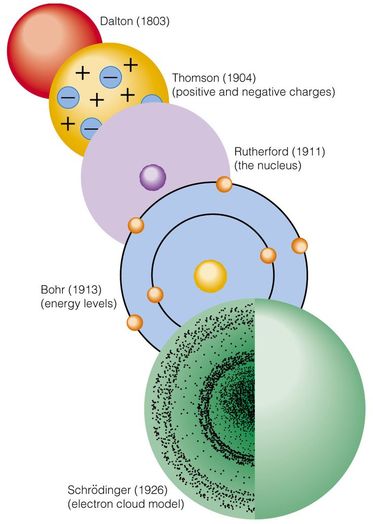
Atomic Theory History: BRIEFLY summarize the 5 models
| |||||||
Chemical Reactions- 109 watch the rest of the video and describe why each one is a chemical reaction.
Physical vs. Chemical Properties Worksheet
| physical_vs_chemical_changes_worksheet.pdf | |
| File Size: | 273 kb |
| File Type: | |
12 Stations Representing 12 Physical Properties
| organizing_matter_lab_stations_results.pdf | |
| File Size: | 50 kb |
| File Type: | |
Lab Safety Quiz - In order to safely participate in class, you need to get at least 2/3 symbols right and all of the questions. Curious about what you got? See below!
|
| ||||||||||||
** Didn't pass? No worries! You can retake this test as many times as necessary.
Lab Safety
|
|
|
Periodic Table Print Out
| periodictablebw2017.pdf | |
| File Size: | 1213 kb |
| File Type: | |